- HOMEGROWN WORLD
- #HGCREATORS
- #HGEXPLORE
- #HGVOICES
- #HGSHOP
- CAREERS
- ABOUT US
- CONTACT US

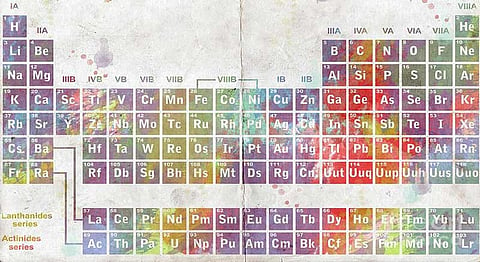
As students, we came up with all kinds of funny mnemonics to memorise the first 20 to 30 elements of the periodic table for science class—and it really was a pain. We have a Russian chemist to thank for the organisation of the elements, which would have been way harder to learn without the proper and organised structure of the table—the first successful periodic table was formulated back in 1869 by Dimitri Mendeleev. His classifications had gaps for elements that were to be discovered in the future. Acknowledging this, he was confident enough in his findings that he used the table he created to make predictions of the atomic weights, densities and oxides of those elements yet to be discovered. His widespread predictions were later realised and held to be true in 1875, 1879 and 1886 when Paul-Émile de Boisbaudran, Lars Nilson and Lemens Winkler, respectively, discovered and named elements that proved Mendeleev correct.
And now, 2016 has started with a lot of excitement for the scientific community as four new elements have been added to the periodic table to finally complete the seventh row. Discovered by scientists from Japan, the United States and Russia—and verified by the International Union of Pure and Applied Chemistry (IUPAC), the highest authority in the respective field—elements 113, 115, 117 and 118 were formally accepted and inducted into the table. As stated by IUPAC in a press release, “claims for discovery of these elements have been fulfilled, in accordance with the criteria for the discovery of elements of the IUPAP/IUPAC Transfermium Working Group (TWG) 1991 discovery criteria.” "To scientists, this is of greater value than an Olympic gold medal," Ryoji Noyori, Nobel laureate in chemistry, told Guardian.
These are the first elements to be included in the table since 2011, when elements 114 and 116 were added. The scientists responsible for the discovery of each element have been invited to name the elements and provide the chemical symbols. A collaboration of Russian-American scientists at the Joint Institute for Nuclear Research in Dubna, Russia, Oak Ridge National Laboratory, Oak Ridge, Tennessee, U.S.A. and Lawrence Livermore National Laboratory in California, U.S.A. produced enough evidence to lay claim on the discovery of elements 115, 117 and 118—temporarily named Ununpentium, Ununseptium and Ununoctium, respectively. Named Ununtrium, element 113’s discovery has been awarded to a team of scientists from the Riken institute in Japan, though the Russians and Americans had also laid claim to that.
IUPAC’s press release explains that the naming of new elements is in the hands of the discoverers. It can be named after a mythological concept, a mineral, a place or country, a property or a scientist. Once named and given a symbol, it’s reviewed by the Inorganic Chemistry Division of IUPAC for consistency, prior historical use and the ease of translatability into various other languages. After it’s accepted, it’s made available for public review for five months, and then the Council, the highest body of IUPAC, takes the final decision on the name of the new chemical elements and their two-letter symbols before the introduction into the periodic table. These new elements do not exist in nature, they’re man-made. They were “discovered by slamming lighter nuclei into each other and tracking the following decay of the radioactive superheavy elements. Like other superheavy elements that populate the end of the periodic table, they only exist for fractions of a second before decaying into other elements,” explains Guardian. The fact that they exist only for a moment made gathering sufficient evidence of their existence very difficult.
IUPAC President Dr. Mark C. Cesa, added to the press release stating, "we are excited about these new elements, and we thank the dedicated scientists who discovered them for their painstaking work, as well the members of the IUPAC/IUPAP Joint Working Party for completing their essential and critically important task."
Meanwhile, we wonder what kind of mnemonics will be used once these four elements get their permanent names.
Words: Sara Hussain
